AI-Generated Video Summary by NoteTube
YouTube Video aaPgsDoZMsU
Overview
This video explores the concept of isotopes, explaining how atoms of the same element can differ in their number of neutrons, leading to variations in mass number. It uses hydrogen and carbon as examples, highlighting carbon-14's role in radiocarbon dating. The discussion then delves into the atomic mass unit (amu) and how atomic weight is calculated as an average of naturally occurring isotopes, illustrated with nitrogen and chlorine. The video transitions to the structure of the atom, discussing the classical model's shortcomings in explaining electron behavior and the inability of atoms to exist if electrons continuously radiated energy. It introduces Niels Bohr's model, which proposes quantized electron orbits and explains the emission and absorption of energy when electrons change these orbits, linking this to atomic spectra and the work of Planck and Einstein.
This summary expires in 30 days. Save it permanently with flashcards, quizzes & AI chat.
Chapters
- •Atoms of the same element can vary in the number of neutrons, which are called isotopes.
- •The mass number (protons + neutrons) distinguishes between different isotopes of an element.
- •Hydrogen has three common isotopes: regular hydrogen, deuterium, and tritium.
- •Carbon has three common isotopes: carbon-12, carbon-13, and carbon-14.
- •Carbon-14 is a radioactive isotope used in radiocarbon dating.
- •Radiocarbon dating compares the ratio of carbon-14 to carbon-13.
- •Stable isotopes (like C-13) remain constant, while radioactive isotopes (like C-14) decay over time.
- •Radioactive decay converts mass into energy, as described by E=mc².
- •The atomic mass unit (amu) is defined as 1/12th the mass of a carbon-12 isotope.
- •Carbon-12 has 6 protons and 6 neutrons, with a defined mass of exactly 12 amu.
- •Atomic weight is the average mass of an element calculated over all its naturally occurring isotopes.
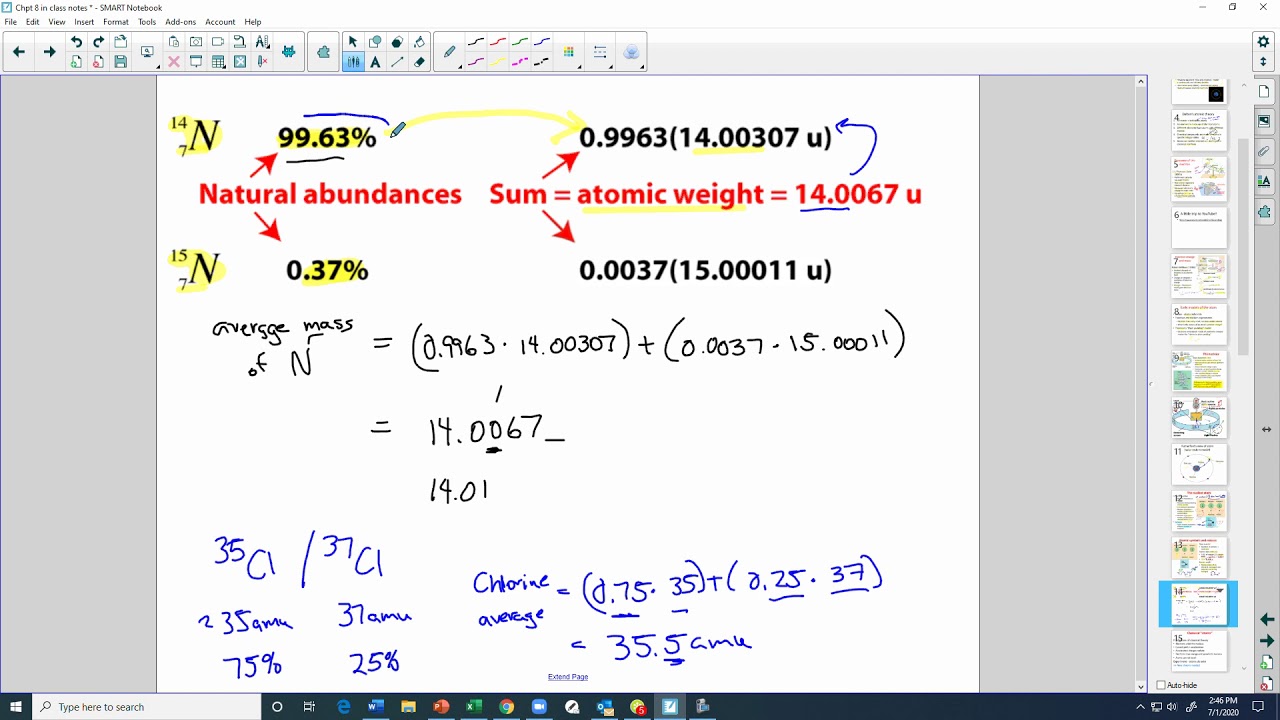
- •Nitrogen has two common isotopes: nitrogen-14 (most abundant) and nitrogen-15.
- •Average atomic mass is calculated by summing the product of each isotope's decimal abundance and its mass.
- •Example calculation for nitrogen shows how its atomic weight (approx. 14.0067) is derived.
- •Different periodic tables may show atomic weights with varying degrees of precision.
- •Chlorine has two common isotopes: chlorine-35 (approx. 75%) and chlorine-37 (approx. 25%).
- •The classical model suggests electrons orbit the nucleus like planets.
- •According to Maxwell's equations, accelerating charges (like orbiting electrons) should emit energy.
- •This emission of energy would cause electrons to spiral into the nucleus, making atoms unstable.
- •Experimental evidence shows atoms are stable, indicating the classical model is incomplete.
- •Niels Bohr proposed a model where electrons exist in specific, allowed orbits.
- •Within these orbits, electrons do not radiate energy.
- •Energy is emitted or absorbed only when an electron transitions between orbits.
- •Max Planck introduced the concept of quantized energy, where photon energy is E=hf (or E=hν).
- •Einstein recognized light as quantized bundles of energy called photons.
- •Exciting an atom causes it to emit light at specific, discrete wavelengths (atomic spectra).
- •This differs from blackbody radiation, which has a continuous energy distribution.
- •Hydrogen's spectrum shows distinct lines, which Bohr's model explains as electron transitions.
- •When an electron moves to a lower energy orbit, it emits a photon with energy equal to the energy difference between the levels.
Key Takeaways
- 1Isotopes are atoms of the same element with different numbers of neutrons, affecting their mass number.
- 2Atomic weight is a weighted average of the masses of an element's naturally occurring isotopes.
- 3The atomic mass unit (amu) is based on the carbon-12 isotope, serving as a standard for atomic masses.
- 4The classical model of the atom fails to explain electron stability and atomic existence.
- 5Niels Bohr's model introduced quantized energy levels for electrons, explaining atomic stability.
- 6Electrons emit or absorb energy (as photons) only when transitioning between allowed energy orbits.
- 7Atomic emission spectra, with their discrete lines, provide evidence for quantized energy levels in atoms.
- 8The work of Planck and Einstein on quantized energy was crucial in developing modern atomic theory.