The Complete Nitrogen Cycle Explained | Microbiology
Microbiology Mantra
Overview
This video explains the crucial nitrogen cycle, a natural process essential for life on Earth. It details how atmospheric nitrogen, unusable by most organisms, is converted into forms that plants and animals can utilize. The cycle involves several key steps: nitrogen fixation (converting N2 gas into ammonia), nitrification (transforming ammonia into nitrites and nitrates), assimilation (uptake by plants and incorporation into organic molecules), ammonification (decomposition of organic matter back into ammonia), and denitrification (returning nitrogen gas to the atmosphere). Microorganisms play a vital role throughout this continuous recycling process, ensuring the availability of this essential element for all living things.
Save this permanently with flashcards, quizzes, and AI chat
Chapters
- Nitrogen is abundant in the atmosphere (78% of air) but exists as a stable N2 gas with a strong triple bond, making it unusable by most plants and animals.
- Nitrogen is a critical component of essential biomolecules like proteins, nucleic acids (DNA/RNA), and ATP, making it vital for all life.
- The nitrogen cycle is Earth's natural recycling system that converts atmospheric nitrogen into usable forms and circulates it through ecosystems.
- Nitrogen fixation is the conversion of atmospheric nitrogen gas (N2) into ammonia (NH3) or related compounds.
- Biological fixation is performed by specialized bacteria using the enzyme nitrogenase, which can break the N2 triple bond.
- Symbiotic bacteria (like Rhizobium in legume root nodules) and free-living bacteria (like Azotobacter) are key players in biological fixation.
- Physical fixation occurs naturally through high-energy events like lightning, which converts N2 into nitrates.
- Industrial fixation, primarily the Haber-Bosch process, uses high temperature and pressure to synthesize ammonia for fertilizers.
- Nitrification is a two-step biological process that converts ammonia/ammonium into nitrites (NO2-) and then into nitrates (NO3-).
- The first step is carried out by ammonia-oxidizing bacteria (e.g., Nitrosomonas), converting ammonia to nitrite.
- The second step is performed by nitrite-oxidizing bacteria (e.g., Nitrobacter), converting nitrite to nitrate.
- These bacteria are chemoautotrophs, using the chemical energy released from these oxidation reactions for their survival.
- Assimilation is the process where plants and microorganisms absorb inorganic nitrogen (nitrates, ammonium) and convert it into organic molecules.
- Plants absorb nitrates and ammonium from the soil and use them to synthesize amino acids, proteins, and nucleic acids.
- Animals obtain organic nitrogen by consuming plants or other animals, incorporating it into their own tissues.
- This step integrates nitrogen into the food web, making it available to higher trophic levels.
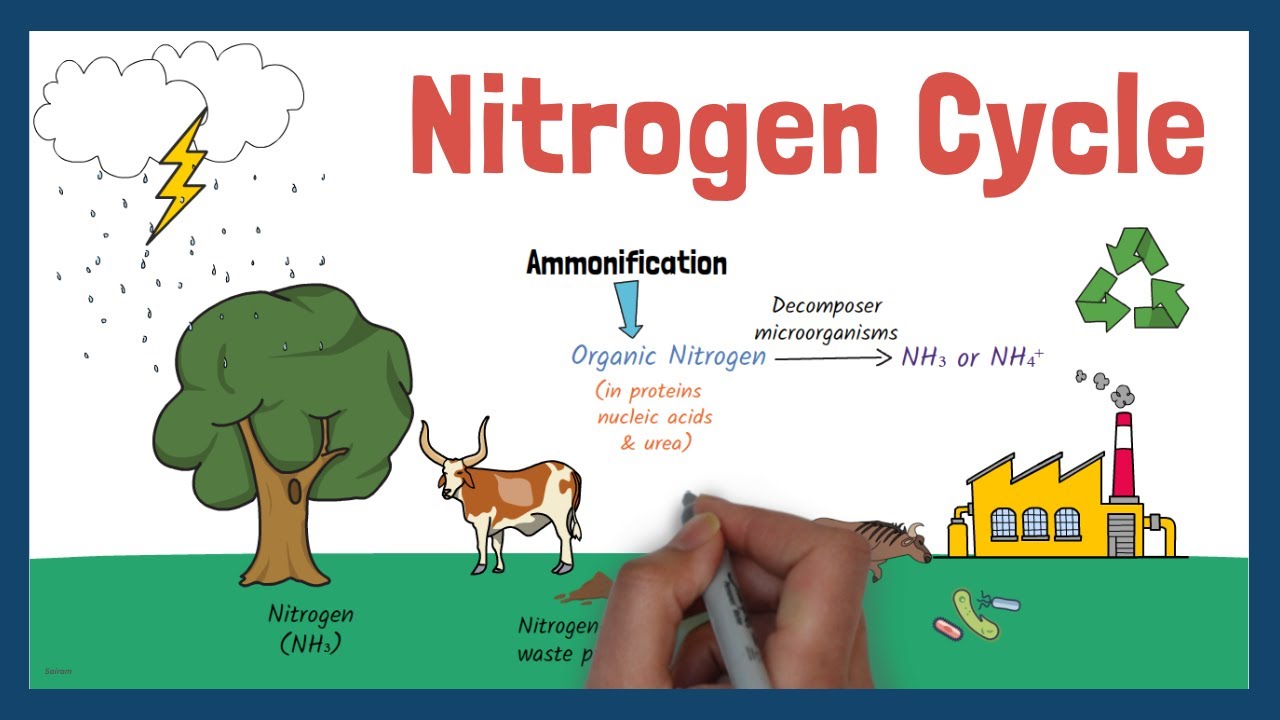
- Ammonification is the decomposition of organic nitrogen (from dead organisms and waste products) back into ammonia or ammonium ions.
- Decomposer microorganisms, primarily bacteria and fungi, carry out this process.
- They break down complex organic molecules like proteins and nucleic acids into amino acids, and then remove amino groups (deamination) to release ammonia.
- This process returns nitrogen to the soil, making it available for plants or further nitrification.
- Denitrification is the microbial process that converts nitrates and nitrites back into nitrogen gas (N2).
- This process occurs primarily under anaerobic (low-oxygen) conditions, such as in waterlogged soils or sediments.
- Denitrifying bacteria use nitrate as an alternative electron acceptor during respiration.
- The process involves a series of reductions: nitrate → nitrite → nitric oxide → nitrous oxide → nitrogen gas.
- This step completes the cycle by returning nitrogen to the atmosphere.
Key takeaways
- Nitrogen is essential for life but exists primarily as unusable N2 gas in the atmosphere.
- Microorganisms are the primary drivers of the nitrogen cycle, performing critical transformations.
- Nitrogen fixation is the essential first step, converting atmospheric N2 into biologically available forms like ammonia.
- Nitrification converts ammonia into nitrates, the most readily absorbed form of nitrogen by plants.
- Assimilation incorporates nitrogen into organic molecules within plants and animals, forming the basis of food webs.
- Ammonification recycles nitrogen from dead organic matter and waste back into the soil.
- Denitrification returns nitrogen gas to the atmosphere, completing the cycle and preventing nitrogen overload in ecosystems.
- Human activities, like industrial fertilizer production (Haber-Bosch process), significantly impact the natural nitrogen cycle.
Key terms
Test your understanding
- Why is atmospheric nitrogen gas (N2) unusable by most plants and animals, and what process makes it available?
- Describe the roles of symbiotic and free-living bacteria in biological nitrogen fixation.
- How does the Haber-Bosch process differ from biological nitrogen fixation, and what is its significance?
- Explain the two main steps of nitrification and the types of bacteria involved.
- What is assimilation, and how do plants and animals obtain nitrogen through this process?
- How does ammonification contribute to the recycling of nitrogen in an ecosystem?
- Under what conditions does denitrification occur, and why is it important for balancing the nitrogen cycle?