AI-Generated Video Summary by NoteTube
YouTube Video e5rZgZm_zOU
0:00
Overview
This video explains the historical experiments and discoveries that led to our current understanding of atomic structure. It begins with Millikan's oil-drop experiment, which determined the charge of an electron and, combined with Thomson's work, its mass. The video then details Rutherford's gold foil experiment, which disproved Thomson's plum pudding model and established the existence of a dense, positively charged nucleus. Finally, it discusses the concepts of atomic number, protons, neutrons, electrons, and isotopes, explaining how these components define elements and their variations.
How was this?
This summary expires in 30 days. Save it permanently with flashcards, quizzes & AI chat.
Chapters
- •Millikan's experiment used charged plates to suspend oil droplets.
- •The experiment measured the charge on individual oil droplets.
- •It determined the fundamental charge of an electron (1.6 x 10^-19 Coulombs).
- •Combined with Thomson's charge-to-mass ratio, the mass of the electron was calculated.
- •The mass of an electron is approximately 9.11 x 10^-31 kg.
- •This mass is very small, about 1/2000th the mass of the lightest atom (hydrogen).
- •This small mass was initially puzzling, as electrons were considered fundamental components of atoms.
- •The discovery confirmed the existence of a lightweight, negatively charged particle: the electron.
- •Following the discovery of the electron, scientists questioned the structure of the atom.
- •Dalton's view of indivisible atoms was challenged.
- •Thomson proposed the plum pudding model: electrons (raisins) embedded in a diffuse positive charge (pudding).
- •This model suggested that the positive charge was spread throughout the atom.
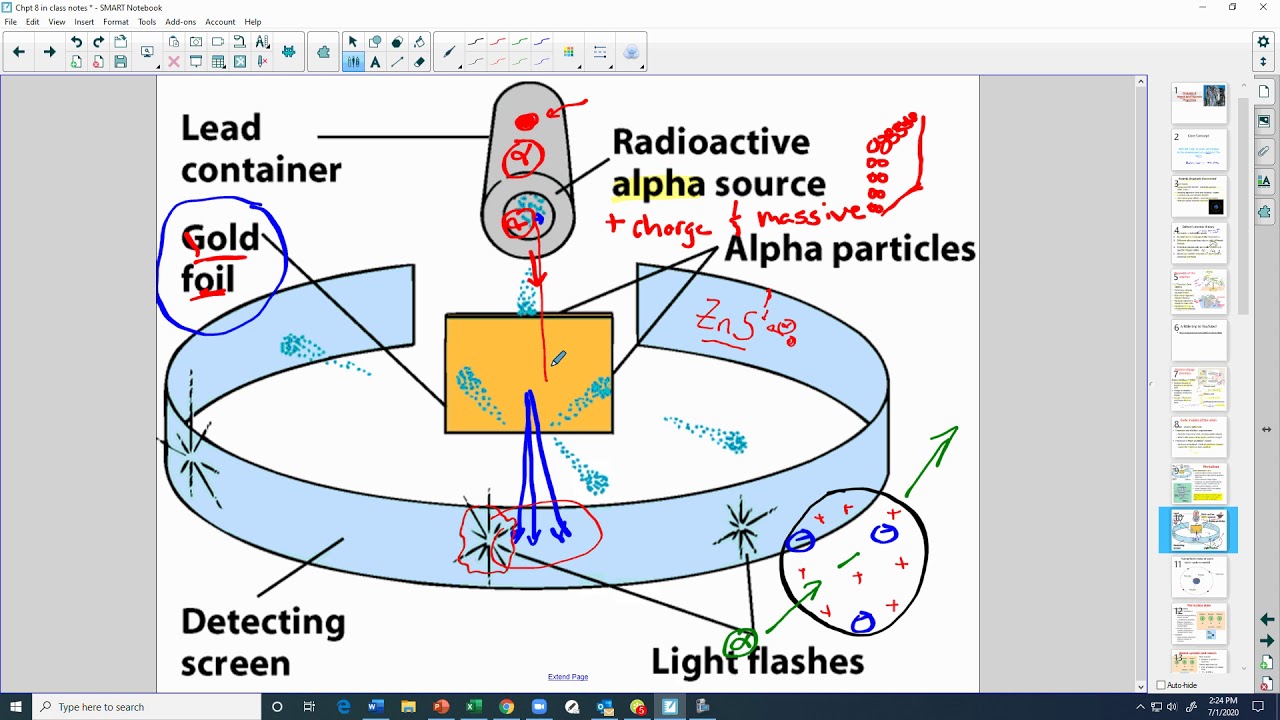
- •Rutherford aimed to test Thomson's plum pudding model using alpha particles.
- •Alpha particles are positively charged and massive.
- •A thin sheet of gold foil was bombarded with alpha particles.
- •Detection involved a zinc sulfide screen that flashed when struck by an alpha particle.
- •Most alpha particles passed straight through the gold foil, as expected.
- •However, some particles were deflected at large angles, and a few even bounced back.
- •This indicated that the positive charge and most of the mass were concentrated in a tiny, dense region: the nucleus.
- •Rutherford's experiment disproved the plum pudding model.
- •Rutherford proposed the nuclear model, with a central nucleus containing protons (positive charge).
- •Electrons orbit the nucleus at a distance.
- •Later, neutrons (neutral charge, significant mass) were discovered by Chadwick, also within the nucleus.
- •This model is often compared to a solar system, with the nucleus as the sun and electrons as planets.
- •The atomic number (number of protons) defines an element.
- •In a neutral atom, the number of protons equals the number of electrons.
- •A charged atom is called an ion.
- •Isotopes are atoms of the same element with different numbers of neutrons.
- •Examples include hydrogen, deuterium, and tritium.
Key Takeaways
- 1Millikan's experiment quantified the charge and mass of the electron.
- 2The electron's mass is minuscule compared to the atom's total mass.
- 3Thomson's plum pudding model proposed a diffuse distribution of positive charge.
- 4Rutherford's gold foil experiment revealed a concentrated, dense nucleus within the atom.
- 5The nuclear model, with protons and neutrons in the nucleus and electrons orbiting, is the basis of modern atomic theory.
- 6The atomic number (number of protons) uniquely identifies an element.
- 7Isotopes are variations of an element differing only in their neutron count.