AI-Generated Video Summary by NoteTube
YouTube Video 64TMApJknZs
Overview
This video introduces the fundamental concepts of electricity and electric force, drawing parallels with gravitational force. It explains the distinction between contact forces and field forces, with gravity and electrostatic forces being prime examples of the latter. The discussion delves into the atomic structure, highlighting protons, neutrons, and electrons as the basis of charge. The video clarifies the nature of positive and negative charges, explaining how like charges repel and opposite charges attract. It introduces the concept of quantization of charge and the unit of charge, the Coulomb. Furthermore, it explores the idea of electric fields as a way to visualize and understand how electric forces act at a distance, contrasting them with gravitational fields. The video also touches upon Coulomb's Law, the mathematical description of electric force, and its similarities and differences with Newton's Law of Universal Gravitation. Finally, it addresses practical applications and problem-solving related to electric forces and fields.
This summary expires in 30 days. Save it permanently with flashcards, quizzes & AI chat.
Chapters
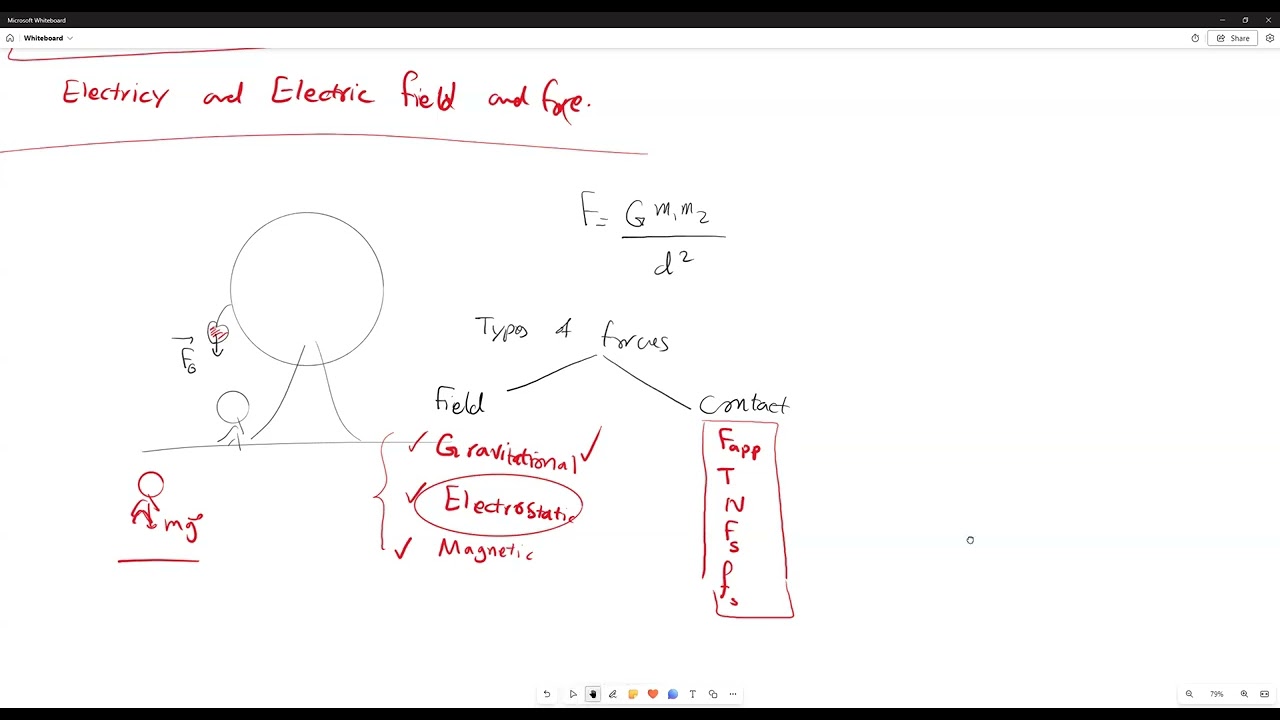
- •Forces can be categorized into contact forces (requiring physical touch) and field forces (acting at a distance).
- •Gravitational force is a field force, as demonstrated by an apple falling from a tree without direct contact.
- •Contact forces include friction, tension, and normal force, all requiring physical interaction.
- •Field forces include gravitational force and electrostatic force, which act across space.
- •Atoms are composed of protons, neutrons, and electrons.
- •Protons carry a positive charge, electrons carry a negative charge, and neutrons are neutral.
- •In a neutral atom, the number of protons equals the number of electrons.
- •The interaction between electrons and protons (attraction) and between like charges (repulsion) led to the concept of two types of charge.
- •Like charges (positive-positive or negative-negative) repel each other.
- •Opposite charges (positive-negative) attract each other.
- •Objects can become charged by gaining or losing electrons through processes like friction (rubbing).
- •Charge is conserved; it cannot be created or destroyed, only transferred.
- •Electric charge is quantized, meaning it exists in discrete multiples of the elementary charge (the charge of an electron or proton).
- •The unit of electric charge is the Coulomb (C).
- •The charge of an electron is approximately -1.60 x 10^-19 C, and a proton is +1.60 x 10^-19 C.
- •Common prefixes like micro (10^-6) and milli (10^-3) are used for smaller charge units.
- •Electric fields are used to visualize the force exerted by charges on other charges in space.
- •The direction of the electric field is defined as the direction of the force on a positive test charge.
- •Electric field lines originate from positive charges and terminate on negative charges.
- •The force on a charge (q) in an electric field (E) is given by F = qE.
- •Coulomb's Law describes the magnitude of the electrostatic force between two point charges: F = k * |q1*q2| / r^2.
- •The electrostatic constant (k) is significantly larger than the gravitational constant (G), making electric forces much stronger than gravitational forces at atomic scales.
- •Unlike gravity, which is always attractive, electrostatic force can be either attractive or repulsive.
- •Electric forces are fundamental to the structure of atoms and molecules, holding them together and forming the basis for most contact forces.
Key Takeaways
- 1Electric forces, like gravity, act at a distance through fields.
- 2Charge is a fundamental property of matter, existing in positive and negative forms.
- 3Like charges repel, and opposite charges attract.
- 4Electric charge is quantized, existing in discrete multiples of the elementary charge.
- 5The unit of electric charge is the Coulomb (C).
- 6Electric fields provide a way to visualize and calculate electric forces.
- 7Coulomb's Law quantifies the electrostatic force between point charges.
- 8Electric forces are significantly stronger than gravitational forces and are responsible for the structure of matter.