AI-Generated Video Summary by NoteTube
Surface Tension of Water, Capillary Action, Cohesive and Adhesive Forces - Work & Potential Energy
The Organic Chemistry Tutor
Overview
This video explains the concept of surface tension in fluids, particularly water. It details how surface tension arises from cohesive forces between molecules, causing fluids to minimize their surface area and form droplets or support light objects. The video quantifies surface tension using the formula gamma = Force / (2 * Length) and demonstrates its calculation with a practical example. It also explores the relationship between temperature and surface tension, noting that higher temperatures decrease surface tension. Finally, the video introduces capillary action, explaining how the interplay between adhesive and cohesive forces determines whether a fluid rises or falls within a narrow tube, using water and mercury as contrasting examples.
This summary expires in 30 days. Save it permanently with flashcards, quizzes & AI chat.
Chapters
- •Surface tension causes water to form droplets and minimize surface area.
- •It allows dense objects, like needles, to rest on the water's surface.
- •Insects can walk on water due to surface tension.
- •Surface tension arises from cohesive forces between fluid molecules.
- •Molecules at the surface experience a net downward force due to attraction to interior molecules.
- •This downward force pulls surface molecules inward, minimizing surface area.
- •A sphere is the shape that minimizes surface area for a given volume.
- •Surface tension is responsible for the spherical shape of small water droplets.
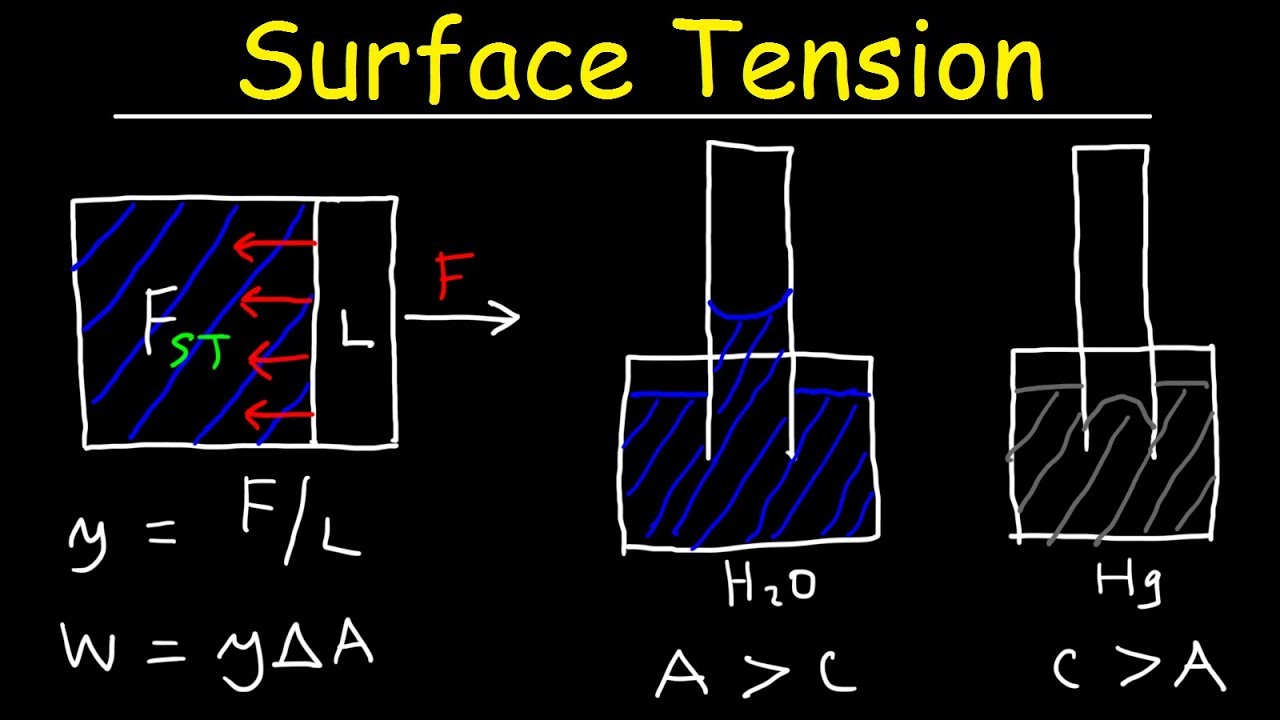
- •Surface tension (gamma) is defined as Force per unit Length.
- •The unit for surface tension is Newtons per meter (N/m).
- •When calculating, the length considered is on both sides of the film, hence 2L.
- •Example calculation: Force = 3.6 x 10^-3 N, Length = 5 cm. Surface tension = 0.036 N/m.
- •Work done to increase surface area is Work = Force x Displacement.
- •Force = gamma * L, and Displacement * L = Area.
- •Therefore, Work = gamma * Area.
- •Increasing surface area increases the potential energy of the fluid, similar to stretching a spring.
- •Increasing temperature decreases surface tension.
- •At 0°C, water's surface tension is 0.076 N/m.
- •At 20°C, it's 0.072 N/m.
- •At 100°C, it's 0.059 N/m.
- •Capillary action is the rise or fall of a fluid in a narrow tube (capillary).
- •It depends on the balance between adhesive and cohesive forces.
- •Water rises in a glass tube because adhesive forces (water-glass) > cohesive forces (water-water).
- •Mercury falls below the surrounding level because cohesive forces (mercury-mercury) > adhesive forces (mercury-glass).
- •Adhesive forces: attraction between different types of molecules.
- •Cohesive forces: attraction between the same type of molecules.
- •Water's meniscus is concave up due to stronger adhesion to glass.
- •Mercury's meniscus is concave down due to stronger cohesion to itself.
Key Takeaways
- 1Surface tension is a property of liquids that arises from cohesive forces, causing the liquid to minimize its surface area.
- 2This phenomenon allows small, dense objects and insects to rest on the surface of water.
- 3Surface tension can be quantified as the force per unit length (gamma = F / 2L) and is measured in N/m.
- 4Work is required to increase the surface area of a liquid, and this work is equal to the surface tension multiplied by the change in area (W = gamma * A).
- 5Increasing the temperature of a liquid generally decreases its surface tension.
- 6Capillary action describes the behavior of liquids in narrow tubes, driven by the interplay of adhesive and cohesive forces.
- 7When adhesive forces are stronger than cohesive forces (e.g., water in glass), the liquid rises.
- 8When cohesive forces are stronger than adhesive forces (e.g., mercury in glass), the liquid level falls below the surrounding surface.