AI-Generated Video Summary by NoteTube
Noble Gas Electron Configurations
chemistryvodcasts
Overview
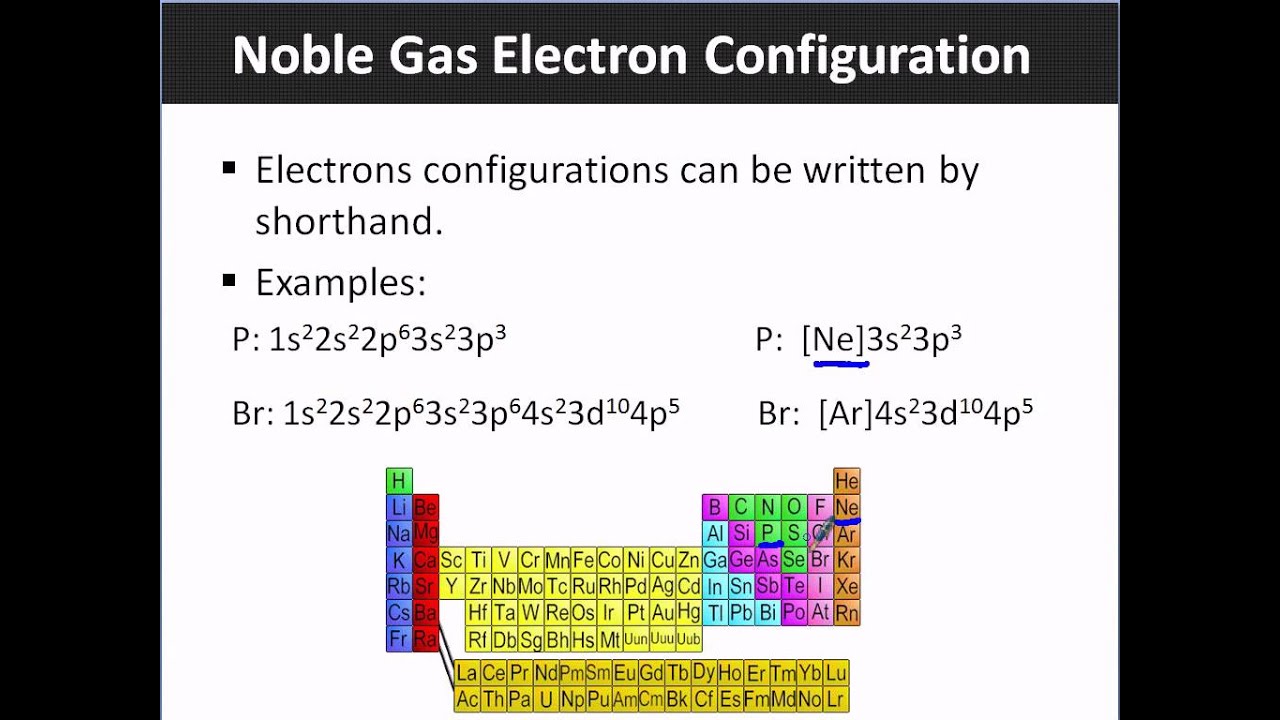
This video explains how to write noble gas electron configurations, a shortcut to simplify writing long electron configurations. It assumes familiarity with standard electron configurations and demonstrates the method using examples like phosphorus and bromine. The core technique involves identifying the noble gas preceding the element in question on the periodic table, writing its symbol in brackets, and then continuing the electron configuration from the next period. This method significantly shortens the notation, making it easier to represent the electron arrangements of elements, especially those further down the periodic table. The video concludes with practice problems for magnesium, iron, and barium, reinforcing the application of this efficient shorthand.
This summary expires in 30 days. Save it permanently with flashcards, quizzes & AI chat.
Chapters
- •Noble gas electron configuration is a shortcut for writing long electron configurations.
- •Assumes prior knowledge of standard electron configurations.
- •Simplifies notation for elements, especially those in lower periods.
- •Locate the element on the periodic table.
- •Identify the most recent preceding noble gas.
- •Write the noble gas symbol in brackets.
- •Continue the electron configuration from the next period.
- •Phosphorus's standard configuration is 1s2 2s2 2p6 3s2 3p3.
- •The preceding noble gas is Neon (Ne).
- •Noble gas configuration: [Ne] 3s2 3p3.
- •Bromine's standard configuration is lengthy.
- •The preceding noble gas is Argon (Ar).
- •Noble gas configuration: [Ar] 4s2 3d10 4p5.
- •Magnesium's preceding noble gas is Neon (Ne).
- •Noble gas configuration: [Ne] 3s2.
- •Iron's preceding noble gas is Argon (Ar).
- •Noble gas configuration: [Ar] 4s2 3d6.
- •Barium's preceding noble gas is Xenon (Xe).
- •Noble gas configuration: [Xe] 6s2.
Key Takeaways
- 1Noble gas electron configuration is a valuable shorthand.
- 2The key is to find the noble gas immediately before the element.
- 3Bracketed noble gas symbol replaces the core electron configuration.
- 4The configuration continues from the period following the noble gas.
- 5This method simplifies complex electron configurations.
- 6Practice applying the method to different elements.
- 7It helps in quickly understanding valence electron arrangements.