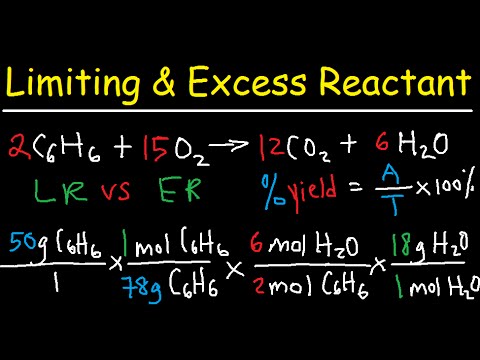
Stoichiometry - Limiting & Excess Reactant, Theoretical & Percent Yield - Chemistry
The Organic Chemistry Tutor
Overview
This video explains the concepts of limiting and excess reactants in chemical reactions, along with how to calculate theoretical yield and percent yield. It demonstrates these principles through two detailed examples: the combustion of propane and the combustion of benzene. The process involves balancing chemical equations, determining the reactant that is consumed first (limiting reactant), calculating the maximum possible product (theoretical yield), and comparing the actual experimental product to the theoretical yield to find the percent yield and percent error. Finally, it shows how to calculate the amount of excess reactant remaining after the reaction is complete.
Save this permanently with flashcards, quizzes, and AI chat
Chapters
- Chemical reactions involve reactants combining to form products.
- The limiting reactant is the substance that is completely consumed first, thus determining the maximum amount of product that can be formed.
- The excess reactant is the substance that is not completely consumed and remains after the reaction stops.
- Understanding these concepts is crucial for predicting reaction outcomes and optimizing chemical processes.
- A balanced chemical equation provides the stoichiometric ratios (mole ratios) between reactants and products.
- To balance combustion reactions, balance carbon, then hydrogen, and finally oxygen.
- One method to identify the limiting reactant is to divide the moles of each reactant by its corresponding coefficient in the balanced equation; the smallest ratio indicates the limiting reactant.
- Alternatively, calculate the theoretical yield of a product from each reactant; the reactant yielding the smaller amount of product is the limiting reactant.
- Theoretical yield is the maximum amount of product that can be formed in a chemical reaction, calculated based on the stoichiometry and the amount of the limiting reactant.
- It represents an ideal scenario where the reaction goes to completion with 100% efficiency.
- Calculations involve converting the moles of the limiting reactant to moles of the desired product using the mole ratio from the balanced equation, and then converting moles of product to grams if necessary.
- Percent yield compares the actual amount of product obtained experimentally (actual yield) to the theoretical yield.
- The formula is: Percent Yield = (Actual Yield / Theoretical Yield) * 100%.
- A high percent yield indicates an efficient reaction, while a low percent yield suggests losses or incomplete reaction.
- Percent error quantifies the difference between the actual and theoretical yields, calculated as: Percent Error = 100% - Percent Yield.
- To find the amount of excess reactant left over, first determine how much of the excess reactant is consumed by the limiting reactant.
- This is done by using stoichiometry, starting with the moles (or grams) of the limiting reactant and converting it to the moles (or grams) of the excess reactant using the mole ratio from the balanced equation.
- Subtract the amount of excess reactant consumed from the initial total amount of excess reactant to find the amount remaining.
- The principles of limiting reactants, theoretical yield, and percent yield apply equally when starting with masses (grams) instead of moles.
- The process requires converting given masses to moles using molar masses, performing stoichiometric calculations using mole ratios, and converting results back to grams if needed.
- The balanced equation for benzene combustion is 2C6H6 + 15O2 -> 12CO2 + 6H2O.
- In this example, 50g of benzene reacts with 160g of oxygen, and 30g of water is collected.
- Using the theoretical yield (34.6g H2O) and the actual yield (30g H2O), the percent yield is calculated as (30g / 34.6g) * 100% = 86.7%.
- To find the excess reactant (O2) remaining, first calculate how much O2 reacts with the limiting reactant (benzene).
- Using 50g of benzene, the amount of O2 consumed is calculated to be approximately 153.1g.
- The remaining excess O2 is the initial amount minus the consumed amount: 160g - 153.1g = 6.9g.
Key takeaways
- The limiting reactant dictates the maximum amount of product possible in a chemical reaction.
- Balanced chemical equations are essential for accurate mole ratio calculations.
- Theoretical yield is a calculated maximum, while actual yield is experimentally measured.
- Percent yield quantifies the efficiency of a reaction by comparing actual to theoretical yield.
- Excess reactants are those not fully consumed, and their remaining amount can be calculated using stoichiometry.
- Stoichiometric calculations can be performed using either moles or masses, requiring conversion steps.
- Understanding these concepts is vital for predicting reaction outcomes and optimizing chemical processes.
Key terms
Test your understanding
- What is the primary role of the limiting reactant in a chemical reaction?
- How does balancing a chemical equation help in identifying the limiting reactant?
- What is the difference between theoretical yield and actual yield?
- How can you calculate the amount of excess reactant left over after a reaction is complete?
- Why is percent yield an important metric in chemistry?