Acids and Bases - Basic Introduction - Chemistry
The Organic Chemistry Tutor
Overview
This video provides a foundational understanding of acids and bases, covering their identification, definitions, and properties. It explains the Arrhenius, Brønsted-Lowry, and Lewis definitions, and how to identify conjugate acid-base pairs. The video also delves into the pH scale, strong vs. weak acids and bases, and their respective behaviors in solution. Finally, it explores acid-base reactions, equilibrium constants (Ka, Kb, Kw), and practical calculations involving pH, pOH, and concentrations, concluding with a review of key concepts and practice problems.
Save this permanently with flashcards, quizzes, and AI chat
Chapters
- Acids often have a hydrogen atom at the beginning of their chemical formula (e.g., HCl, HF).
- Bases typically contain a hydroxide ion (e.g., NaOH, KOH).
- A hydrogen bonded to a nonmetal usually indicates an acid, while a hydrogen bonded to a metal can indicate a base (e.g., NaH).
- Acids tend to release H+ ions (protons), while bases often involve OH- ions or accept protons.
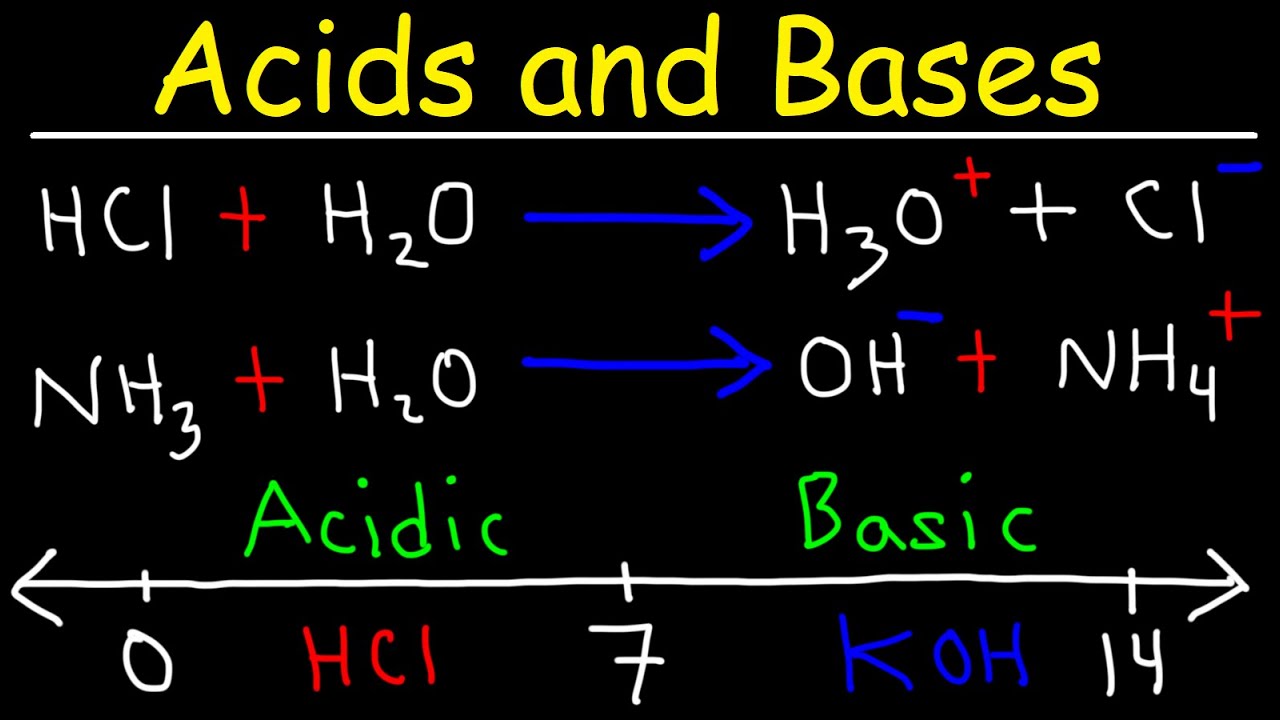
- Arrhenius definition: Acids produce H+ ions in solution, and bases produce OH- ions.
- Brønsted-Lowry definition: Acids are proton (H+) donors, and bases are proton acceptors.
- In a Brønsted-Lowry reaction, the acid forms a conjugate base, and the base forms a conjugate acid.
- Lewis definition: Acids are electron pair acceptors, and bases are electron pair donors.
- The pH scale measures the acidity or basicity of a solution, typically ranging from 0 to 14.
- A pH less than 7 is acidic, pH equal to 7 is neutral, and pH greater than 7 is basic.
- pH is calculated as the negative logarithm of the hydronium ion (H3O+) concentration: pH = -log[H3O+].
- pOH is calculated as the negative logarithm of the hydroxide ion (OH-) concentration: pOH = -log[OH-].
- The relationship pH + pOH = 14 (at 25°C) allows conversion between pH and pOH.
- Strong acids and bases ionize completely in water, forming strong electrolytes.
- Weak acids and bases only partially ionize, forming weak electrolytes.
- Common strong acids include HCl, HBr, HI, HNO3, H2SO4, and HClO4.
- Strong bases are typically soluble metal hydroxides like NaOH and KOH.
- The strength of oxyacids increases with the number of oxygen atoms.
- Reactions involving strong acids/bases use a single arrow (→) to show complete dissociation.
- Reactions involving weak acids/bases use a double arrow (⇌) to indicate a reversible equilibrium.
- The acid dissociation constant (Ka) quantifies the strength of a weak acid; a higher Ka means a stronger acid.
- The base dissociation constant (Kb) quantifies the strength of a weak base; a higher Kb means a stronger base.
- Water can act as both an acid and a base (amphoteric), undergoing autoionization (Kw = [H3O+][OH-]).
- Acids typically taste sour and turn blue litmus paper red.
- Bases typically taste bitter and feel slippery, turning red litmus paper blue.
- Strong acids have high Ka values and low pKa values; strong bases have high Kb values and low pKb values.
- There is an inverse relationship between acid strength and the strength of its conjugate base.
- Ka * Kb = Kw and pKa + pKb = 14 (at 25°C) relate acid and base strengths.
Key takeaways
- Acids and bases can be identified by their chemical formulas and defined by their behavior in donating/accepting protons or releasing ions.
- The pH scale quantifies acidity and basicity, with calculations based on ion concentrations.
- Strong acids and bases dissociate completely, while weak ones only partially dissociate, affecting solution conductivity and reactivity.
- Acid-base reactions often reach an equilibrium state, described by dissociation constants (Ka and Kb).
- Water is amphoteric and undergoes autoionization, with its equilibrium constant (Kw) linking H3O+ and OH- concentrations.
- There are inverse relationships between acid strength and conjugate base strength, and between Ka and pKa.
- Understanding these concepts is crucial for predicting and controlling chemical reactions in various fields.
Key terms
Test your understanding
- How does the Brønsted-Lowry definition of an acid differ from the Arrhenius definition?
- What is the relationship between the pH of a solution and the concentration of hydronium ions?
- Why do strong acids form strong electrolytes, while weak acids form weak electrolytes?
- How can you determine if a substance is amphoteric based on its chemical structure or behavior?
- What is the relationship between the Ka of an acid and the Kb of its conjugate base?